|
DICTIONARY
? »
|
On the welcome page of the dictionary, four major sections are shown: Normal tissues, Cancer, Cell structure and Protein expression. Below the image of each section are links to introductory texts for i) normal tissue histology, ii) hallmarks of cancer, and iii) cell structure overview. For the cancer-section there is also a link to current cancer statistics (incidence, survival, etc) for Sweden and the rest of the world. Below the Protein expression section there is a list of genes with links to examples of different protein expression patterns. Within each section there are direct links to histology descriptions of different tissue types and tumor forms as well as descriptions of cell structures.
For the 'Tissue & cell types' and 'Tumors' sections, tissue-slides stained with hematoxylin and eosin (HE) are shown at three different levels of magnification. On the top level, an overview of the whole tissue-sample is shown with boxes in black indicating where zoomed-in representative parts of the tissue are available for viewing. Clicking on these boxes will zoom in on that part to show tissue structures, cells and features in greater detail. Throughout these sections, arrows indicate relevant tissue structures, cell-types and other features.
For the 'Cell structure' section, immunofluorescent images of formaldehyde-fixed cell lines are shown. The various cell structures that are demonstrated are always shown in the green channel using an antibody found in the Human Protein Atlas. The antibody name is linked to the subcellular location summary page of the target gene. The other channels: nucleus, microtubules and endoplasmic reticulum, are always shown in the blue, red and yellow channels, respectively. The channels can be toggled on and off by clicking on the respective coloured button above the image. When applicable, the immunofluorescent images are complemented by immunohistochemically stained cells where the location of the particular cell structure is shown in brown.
For the 'Protein expression' section, tissue slides representing both normal and cancer tissue have been stained using immunohistochemistry to visualize various protein expression patterns. Antibodies have been selected to demonstrate the expression patterns of well-known proteins and to reflect antibodies used in clinical diagnostics to determine the nature of a given cancer. For certain antibodies the corresponding protein expression pattern is shown in both normal and cancer tissues. For each exemplified protein there is a short descriptive text including also the clinical usefulness of the corresponding antibody.
A common feature for all sections is that a general descriptive text about the tissue, tumor-type, cell structure or protein/antibody is provided when browsing a particular topic.
|
Female, 69 years, moderately differentiated adenocarcinoma, Stage II (T3, N0, M0), cancer invading bowel wall
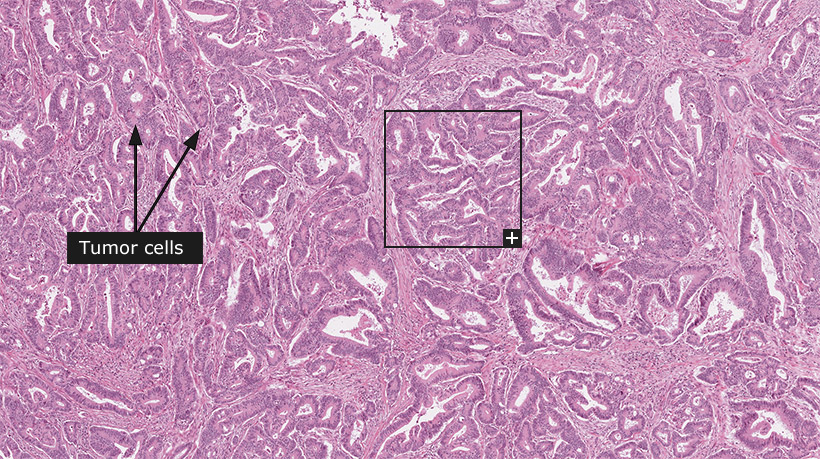
Colorectal cancer
Colorectal cancer (CRC) is the second most common cancer in the industrialized world, accounting for approximately one million new cases each year. Incidence rates correlate with meat consumption and migration studies reinforce the importance of environmental factors. The overall mortality is approximately 50%. The surgical stage at diagnosis is the most important factor for predicting patient outcome, with five year survival rates of more than 90% for stage I disease and less than 10% for stage IV disease.
The surgical stage represents a classification system based on the extent and depth of tumor growth. Stage I CRC shows invasive grown into the anatomical layers of the colon, but the tumor is not spread outside the colon wall or into regional lymph nodes. Stage II CRC demonstrates extended growth through the outer layer of the colon (peritoneum) and may have extended into nearby organs, but has not spread to any lymph node. Stage III CRC have spread to nearby lymph nodes but not yet metastasized to distant sites in the body. Finally, in Stage IV CRC the tumor has spread to distant organs such as the liver, lungs, or other sites. The Dukes classification is an older and less complicated staging system that predates the TNM system, and translates so that Duke A= Stage I, Duke B= Stage II, Duke C= Stage III and Dukes D= Stage IV.
CRC is considered to develop through a multi-step process, originating from normal colon epithelium that develops into precursor lesions termed adenomas. Adenomas can subsequently further progress into invasive CRC with metastatic potential. Spread of CRC occurs by direct growth through the bowel wall and through invasion of lymphatic and venous channels. The most common sites for metastases are regional lymph nodes and the number of lymph node metastases influences prognosis. The liver is the most common distant site for CRC metastases.
The vast majority of CRC are adenocarcinomas, with less than 10% of the cancers being distinguished by an abundant secretion of mucin. The tumors are classified according to the degree of morphological differentiation into well, moderately and poorly differentiated. About 80% are well or moderately differentiated with a growth pattern consisting of tumor cells that form irregular glandular structures present at different layers of the bowel wall. Poorly differentiated CRC show no, or only hinted, glandular formation. Overall poor differentiation with a diffuse infiltrative growth pattern is associated with poor prognosis, although stringent classification systems based on morphological features are lacking. Treatment decisions are thus based on the surgical stage without consideration to morphological characteristics. In addition to adenocarcinomas, endocrine tumors can also arise within the colorectal mucosa. Squamous and adenosquamous tumors are exceedingly rare.
In addition to the diagnostic procedure, which is based on microscopical examination of a tumor specimen, immunohistochemistry can be used to determine a colorectal origin of a metastasis or to visualize the spread of tumor cells in surrounding tissues. Antibodies that show high sensitivity and specificity for tumors of colorectal origin include Cytokeratin 20, CDX-2, SATB2 and Cadherin-17. Chromogranin-A antibodies can be used to distinguish endocrine tumors in the bowel from common adenocarcinomas.
Recently new targeted drugs have been implemented into the treatment of patients with advanced colorectal cancer. Epidermal growth factor receptor (EGFR) is commonly expressed in colorectal tumors and monoclonal antibodies inhibiting EGFR demonstrate clinical efficacy in patients with tumors that do not harbor downstream activating KRAS mutations. Today KRAS mutation status is analyzed routinely before starting anti-EGFR treatment.
Normal tissue: Colon, Rectum
|
|
|
|
|
|
|
|
Protein expression
|

|
SATB2, colorectal cancer
|

|
CDX2, colorectal cancer
|

|
KRT20, colorectal cancer 1, adenocarcinoma
|
|
KRT20, colorectal cancer 2, adenocarcinoma
|

|
CDH1, colorectal cancer
|

|
TP53, colorectal cancer 1, adenocarcinoma
|

|
KRT20, colorectal cancer 3, adenocarcinoma
|

|
TP53, colorectal cancer 2, adenocarcinoma
|

|
VIM, colorectal cancer
|

|
PECAM1, colorectal cancer
|

|
MKI67, colorectal cancer 1, adenocarcinoma
|

|
MKI67, colorectal cancer 2, adenocarcinoma
|

|
MKI67, colorectal cancer 3, adenocarcinoma
|
|
|
|
|